Exsolution of metal nanoparticles from SrTiO3 is driven by raising the Fermi level
A Single Model for the Thermodynamics and Kinetics of Metal Exsolution from Perovskite Oxides
2024/08/12 by AK/df
A new study performed at RWTH Aachen and published in the Journal of the American Chemical Society highlights the essential role of the Fermi level for the solubility of dopants and its role in material processing.
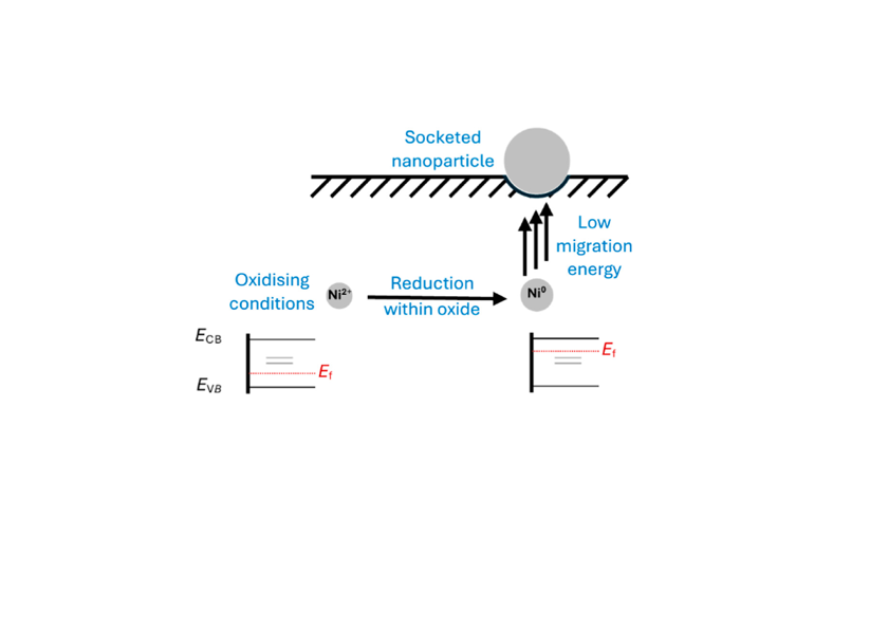
Exsolution is a modern way to introduce catalytically active metal nanoparticles at surfaces. Computational studies of the thermodynamics and kinetics of defects performed at RWTH Aachen, initiated and guided by key FLAIR concepts, revealed that the exsolution of Ni nanoparticles from Ni-doped SrTiO3 is directly related to changes in the Fermi level of the material. Reduction of the material introduces oxygen vacancies and raises the Fermi level. This results in a reduction of the Ni2+ dopants within the oxide to Ni0, species that are unstable in the compound and thus expelled to the surface. The results, which have been published in the Journal of the American Chemical Society, highlight the essential role of the Fermi level for the solubility of dopants and its role in material processing.




